By Valéria Credidio / LAIS/UFRN Communication Advisor
Translation: NRI/LAIS
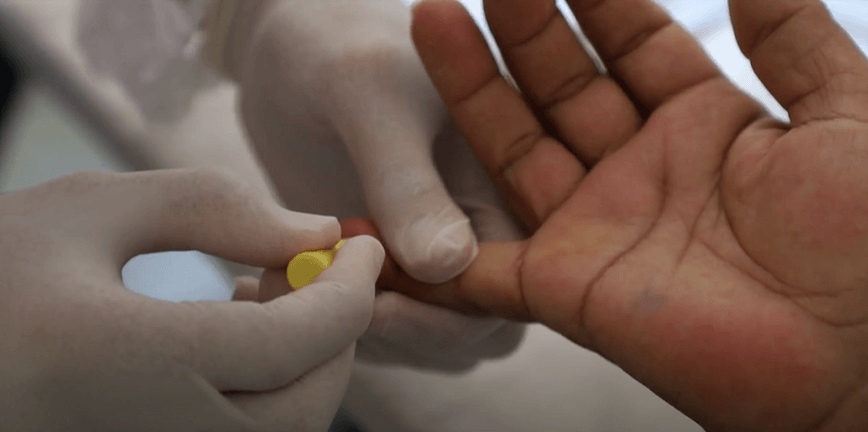
More than 12 million people are infected annually with syphilis worldwide. The data are from the World Health Organization and point to a silent pandemic of one of the oldest sexually transmitted infections of humanity. Implemented since 2017, the “Syphilis No” Project has been acting strongly to transform this scenario. Among the various actions is the development of a test with the potential to perform the diagnosis more effectively and at a lower cost, facilitating the use in the basic health network and fully integrated with the technological ecosystem of the Ministry of Health.
The test is the result of another research conducted by the Laboratory for Technological Innovation in Health (LAIS/UFRN) in partnership with Johns Hopkins University, Baltimore, USA, and the University of Coimbra, Portugal. All the research work was validated, recently, through the publication of the scientific article “Development of a method based on cyclic voltammetry for the detection of antigens and antibodies as a new strategy for the diagnosis of syphilis” (https://pubmed.ncbi.nlm.nih.gov/36498280/), in the International Journal of Environmental Research and Public Health, an international journal of relevance in health research.

Through an innovative methodology, the new syphilis diagnostic test can also be used to detect other sexually transmitted infections (STI), such as HIV. Another important aspect concerns the cost-benefit in relation to traditional methods, facilitating its use in primary care. According to Professor Lucio Gama, a researcher at Johns Hopkins University, current diagnostic tests for syphilis rely on reagents and machines that are expensive and must remain in a laboratory. “The advantage of this new method is portability and the possibility of being used for the diagnosis of other diseases,” he said, stressing that the method used to develop the test has the ability to revolutionize the detection tests for both pathogens and antibodies for various diseases.
The publication of the article is the validation, by the scientific community, of the entire process studied and elaborated by the researchers who are expanding the recruitment of new patients for testing. “The next steps are adjustments to assess the sensitivity and specificity of the test and the preparation of the dossier requesting the registration of this new device with Anvisa,” explained LAIS researcher Leonardo Lima. Besides the registration at Anvisa, the test also has an international patent application.
In the opinion of the General Coordinator of Sexually Transmitted Infections Surveillance, Angélica Espinosa, the results of this article are important because they bring good perspectives for the control of syphilis and congenital syphilis. “The development of new technologies for an accurate diagnosis of syphilis cases is an important tool for infection control because it allows an earlier diagnosis and adequate management of the cases. Having an easy-to-perform, the low-cost rapid test will be a major advance for the public policy that deals with the elimination of vertical transmission of syphilis in our country,” he argued.
Contributions
Much more than a scientific research, the new test for syphilis is an advance in terms of public policies for Brazil. According to the executive director of LAIS, professor Ricardo Valentim, this new testing methodology is an important milestone in the qualification of the diagnosis of this sexually transmitted infection, not only in Brazil but in the world, since the screening methods for syphilis are still insufficient. “The lack of tests generates several problems for tracking, for care, for the elimination of vertical transmission of syphilis, when the mother transmits the STI to the baby during pregnancy or at the time of delivery,” explained the LAIS director.
Another significant contribution, pointed out by Valentim, is the qualification of the diagnosis, bringing a more accurate result. “When incorporated into Brazilian Health System, this test will be available primarily in primary health care, facilitating the integration of primary care with health surveillance, a dominant factor in eliminating congenital syphilis in Brazil.
Valentim also highlighted another essential contribution related to case management. The new test allows interoperability with the existing health information system of the Ministry of Health and with the SALUS* platform itself, also developed by LAIS. Thus, the Ministry of Health and state and municipal health departments will be able to have the results of these tests in real-time. “The data will be linked to the patient’s medical records, which will improve all the follow-up and case management of the patients. Furthermore, the test allows us to apply the concept of segment monitoring, i.e., to look at the entire treatment, with the proposal to monitor the treatment, as well as the evolution of the patient’s clinical picture regarding syphilis – this represents a considerable advance for Brazil.
Partnership
For the development of the test, LAIS counted on important partnerships. One of them was Johns Hopkins University, an American institution with internationally recognized quality.
For Professor Lúcio Gama, also from the National Institutes of Health, in the United States, the partnership with LAIS and the Brazilian researchers is an excellent opportunity for new discoveries. “Science is a collaborative process. It is important to exchange knowledge between different cultures and countries so that problems can be seen with different eyes. I was very impressed with LAIS when I visited the lab facilities. The Brazilian wit and creativity are impressive. Here we join the qualities of Brazilians with the scientific tradition of an entity as old as Johns Hopkins. And this is the success we are seeing.
Another important partnership is with the Brazilian Medical Devices Industry Association (Abimo). For the superintendent of Abimo, Paulo Fraccaro, it is necessary to recognize the importance of LAIS for developing Brazilian health as an international reference in technology and innovation – including studies and evidence published in renowned journals. For Fraccaro, the laboratory provides a very high-quality service to the population. “The LAIS differential is not limited to scientific research. It conducts the study but follows up on the project until it can, in fact, reach the market. This rapid test for Syphilis detection is an excellent example”, he argued.
For Abimo’s superintendent, every Brazilian should be proud of having a laboratory that is concerned with developing quality and cost-effective products and solutions to be incorporated into the Brazilian Health System (SUS), responsible for the exclusive care of 75% of our people.
Fraccaro also pointed out that LAIS is a showcase of the high level of knowledge and competence of the scientific class. “As representatives of the national medical devices industry, we partner with LAIS to bring the laboratory’s scientific teams closer to the R&D teams of Brazilian manufacturers. With this, we are sure that we will be able to further revolutionize the health innovation scenario in Brazil with many new projects capable of assisting in the delivery of safe and quality care for all,” he concluded.
How to cite
Barros GMC, Carvalho DDA, Cruz AS, Morais EKL, Sales-Moioli AIL, Pinto TKB, Almeida MCD, Sanchez-Gendriz I, Fernandes F, Barbalho IMP, Santos JPQ, Henriques JMO, Teixeira CAD, Gil P, Gama L, Miranda AE, Coutinho KD, Galvão-Lima LJ, Valentim RAM. Development of a Cyclic Voltammetry-Based Method for the Detection of Antigens and Antibodies as a Novel Strategy for Syphilis Diagnosis. Int J Environ Res Public Health. 2022 Dec 3;19(23):16206. doi: 10.3390/ijerph192316206. PMID: 36498280; PMCID: PMC9738993.
*Salus – Epidemiological Surveillance Monitoring System, implemented in municipalities of RN, MS, SP, RJ, BA and PE.